Our Research
High Entropy Materials
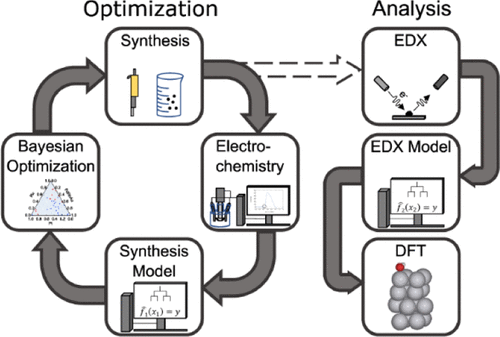
High Entropy Materials as a Platform for Catalyst Discovery
High entropy materials provide a powerful platform for electrocatalyst discovery by combining multiple elements in a single, compositionally complex solid, thereby generating a broad distribution of surface atomic environments. In electrocatalysis, this results in a spectrum of adsorption energies for reactants and key intermediates that ultimately governs catalytic activity. By smoothly varying the composition, we tune the probability of forming surface sites with near-optimal binding under applied electrochemical potential.
Combined with medium-throughput nanoparticle synthesis and machine-learning-based analysis, this approach enables systematic exploration and exploitation of the composition space, understood here as all possible material combinations, including but not limited to classical high entropy materials.
More recently, our work has shifted toward multi-objective optimization using Pareto analysis to identify the set of best achievable trade-offs between competing performance metrics such as activity and stability. Using this framework, we investigate key energy-conversion reactions including the oxygen reduction reaction (ORR), oxygen evolution reaction (OER), CO oxidation, and related processes.
- Journal of the American Chemical Society 148, 4815-4825, 2026
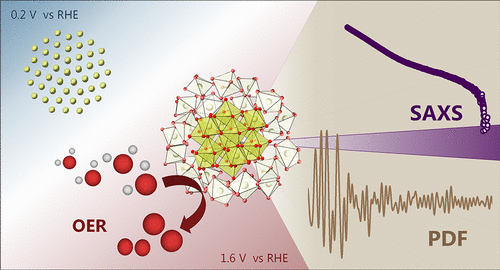
In situ and Operando Investigations of Complex Materials
Establishing structure–activity relationships has long been a central objective in catalysis research. A major challenge, however, lies in the fact that catalyst structures often evolve under reactive conditions, particularly in electrocatalysis. As a result, it is essential to determine catalyst structures in situ or operando, that is, directly under reaction conditions. Only by resolving the atomic arrangement in the working state can we elucidate the mechanisms that govern material properties and catalytic performance.
In addition, we employ in situ techniques to investigate the synthesis of catalyst materials. Monitoring how materials form and evolve during synthesis is crucial for understanding the underlying formation mechanisms. Such insights enable us to rationally control and direct the synthesis process, ultimately facilitating the design of new materials with tailored properties.
Our research primarily employs X-ray–based techniques, including absorption spectroscopy and scattering methods, to probe the atomic structure of catalysts. These studies are conducted in close collaboration with the research groups of Prof. Rebecca Pittkowski, Prof. Kirsten Jensen, and Prof. Mehtap Oezaslan.
- Monitoring the Structural Changes in Iridium Nanoparticles during Oxygen Evolution Electrocatalysis with Operando X-ray Total Scattering; Journal of the American Chemical Society 146 (40), 27517-27527, 2024
- J Schröder, RK Pittkowski, I Martens, R Chattot, J Drnec, J Quinson, JJK Kirkensgaard,;
Synthesis of Improved Catalysts
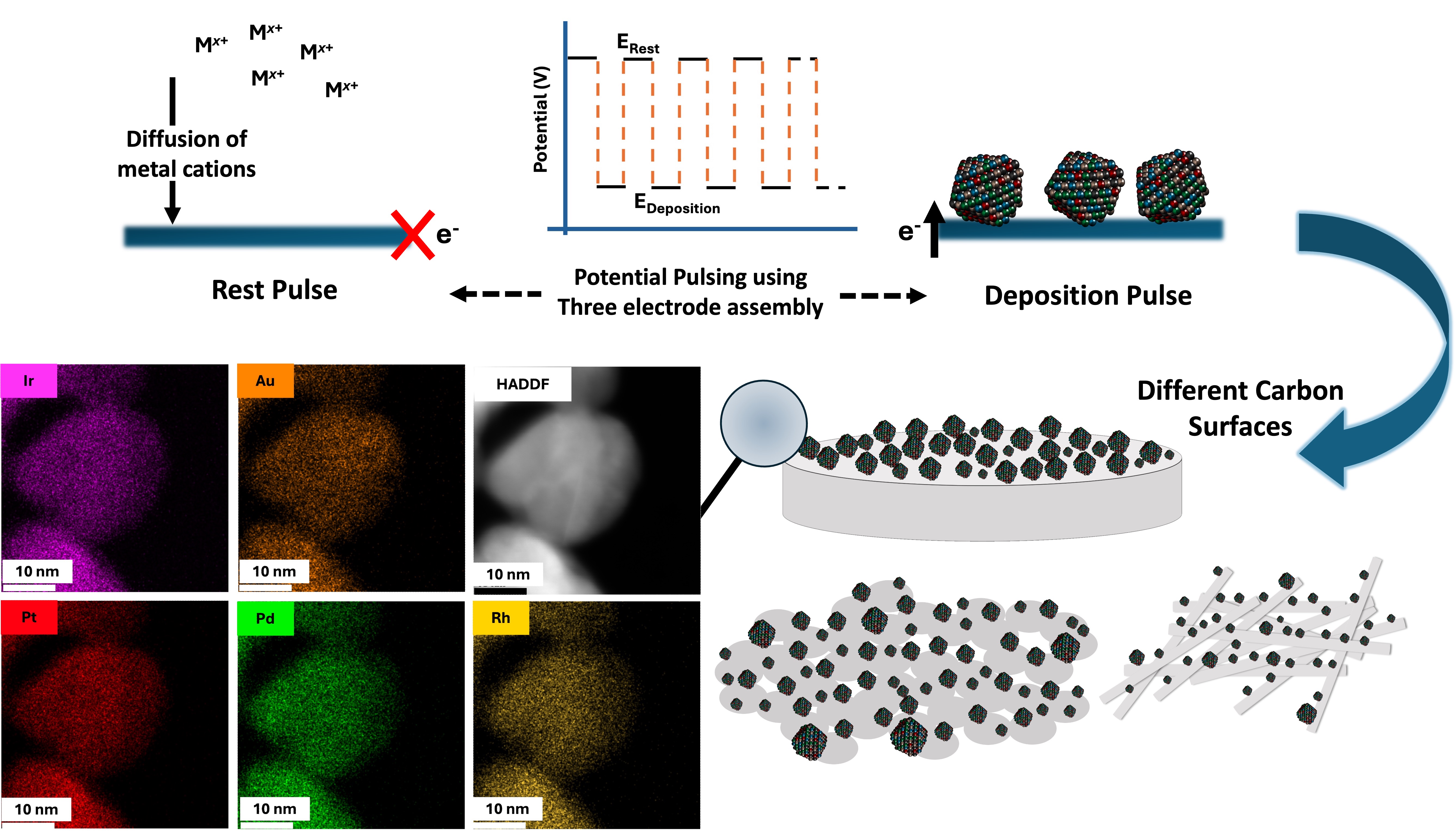
Electrodeposition Approach
We developed a medium-throughput electrodeposition strategy for synthesizing high-entropy alloy (HEA) noble metal nanoparticles on conductive supports. The approach employs aqueous electrolytes and operates at high overpotentials to achieve mass transport–controlled deposition, thereby decoupling the process from the individual equilibrium potentials of the constituent elements.
Hydrogen evolution, which can interfere with alloy formation, is effectively suppressed by applying a pulsed deposition mode and maintaining a mildly acidic pH. The method is rapid, versatile, and allows precise compositional tuning while preserving a homogeneous elemental distribution within the nanoparticles.
- D Gautam, GKH Wiberg, J Quinson, D Wang, CM Clausen, R Rohde, R Klemmt, J Rossmeisl, E Drath Bøjesen, ; Noble Metal‐Based High Entropy Alloy Nanoparticles Prepared by Pulsed Electrodeposition: An Approach for Medium Throughput Studies ; Small Structures 2026, 7(1), e202500666,
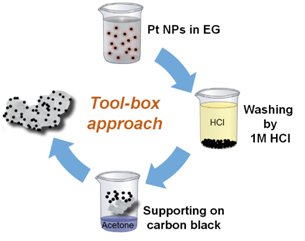
Colloidal Toolbox Approach
We developed a “tool-box” synthesis approach based on “surfactant-free” colloidal nanoparticles in alkaline ethylene glycol. The key advantage over conventional synthesis approaches is that the catalyst properties (particle size, metal loading, support type, etc.) can be changed and optimized independent of each other. Therefore, the approach is ideally suited to systematically investigate complex phenomena, such as the particle size or the particle proximity effect of catalytic processes. This work is mainly done in close collaboration with the group of Dr. Sebastian Kunz and Prof. Jonathan Quinson.
Incipient Wetness Impregnation Approach
We employ a conventional incipient wetness impregnation approach to synthesize high-entropy alloy (HEA) nanoparticles supported on high-surface-area carbon materials. This method is scalable and well established in industrial catalyst production.
By carefully optimizing the synthesis parameters, single-phase HEA nanoparticles can be reliably obtained. Furthermore, subsequent thermal treatment enables the formation of high-entropy intermetallic nanoparticles, providing access to structurally ordered multicomponent materials.
- N Schlegel, S Punke, CM Clausen, U Friis-Jensen, AF Sapnik, D Stoian, O Aalling-Frederiksen, D Gautam, J Rossmeisl, RK Pittkowski, , KMØ Jensen; Tracking the Formation of High Entropy Solid Solutions and High Entropy Intermetallics by In Situ X-ray Diffraction and Spectroscopy ; Chemistry of Materials 37 (3), 939-953, 2025

Support-free, Nanoporous Thin Film Catalysts Prepared by Magnetron Sputtering
We investigate nanoporous metal alloy films as support free catalysts. The key point is to combine a high specific activity, as known for bulk alloys, with high surface area and a porous structure for reactant transport. The films are prepared in the group of Dr. G. Sievers and Dr. Volker Brüser at the INP Greifswald by alternating magnetron sputtering of a noble and a non-noble component and subsequent acid leaching of the non-noble component. We test the catalytic performance as well as the stability of the nanoporous metal alloy films.
The technology led to a spin-off at INP Greifswald, the company Elementarhy
- GW Sievers, AW Jensen, J Quinson, A Zana, F Bizzotto, M Oezaslan, A Dworzak, JJK Kirkensgaard, TEL Smitshuysen, S Kadkhodazadeh, M Juelsholt, KMØ Jensen, K Anklam, H Wan, J Schäfer, K Čépe, M Escudero-Escribano, J Rossmeisl, A Quade, V Brüser, ; Self-supported Pt–CoO networks combining high specific activity with high surface area for oxygen reduction; Nature materials 20 (2), 208-213, 2021
Development of Improved Experimental Instrumentation
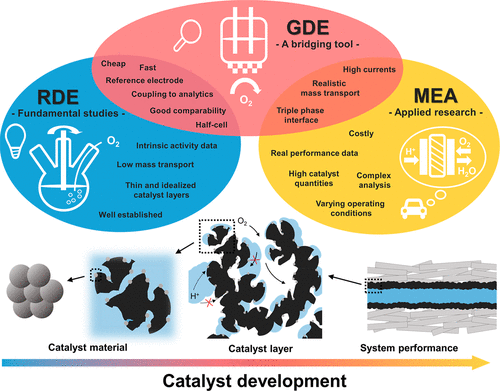
The Gas Diffusion Electrode (GDE) setups as a brigding tool in Electrocatalysis
Gas diffusion electrode (GDE) setups enable the evaluation of electrocatalysts under fuel-cell- and electrolyzer-relevant three-phase conditions, where gas, ionomer, and solid catalyst are in direct contact, while retaining the flexibility of a three-electrode electrochemical half-cell. By allowing the use of realistic catalyst layers, operation at high current densities, and independent control over gas composition, electrolyte, and temperature, the GDE approach effectively bridges the gap between rotating disk electrode (RDE) measurements and full membrane-electrode assemblies (MEAs). In our group, GDE systems have been applied to study the oxygen reduction (ORR), oxygen evolution (OER), and CO2 reduction reactions, including the development of specialized cells for operando X-ray characterization. Most recently, we have advanced this platform by implementing pressurized GDE setups that enable operation at elevated temperatures and pressures, further extending the relevance to practical energy-conversion conditions.
- ; Gas diffusion electrode setup for catalyst testing in concentrated phosphoric acid at elevated temperatures;
- M Inaba, A Westergaard Jensen, GW Sievers, M Escudero-Escribano, A Zana; Benchmarking high surface area electrocatalysts in a gas diffusion electrode: measurement of oxygen reduction activities under realistic conditions ; Energy & environmental science 11 (4), 988-994, 2018
- Benchmarking fuel cell electrocatalysts using gas diffusion electrodes: inter-lab comparison and best practices;
- Benchmarking proton exchange membrane fuel cell cathode catalyst at high current density: A comparison between the rotating disk electrode, the gas diffusion electrode and differential cell;

The Gas Diffusion Electrode (GDE) Setup for Operando Measurements
We developed a gas diffusion electrode (GDE) cell designed for operando investigations of electrocatalytic processes. Building on our established GDE architecture, the cell has been specifically adapted for operando and in situ studies, with particular emphasis on X-ray scattering experiments.
The optimized design enables the minimal sample–detector distances required for comprehensive X-ray total scattering measurements, thereby facilitating high-quality structural characterization under realistic reaction conditions.
- GKH Wiberg, RK Pittkowski, S Punke, O Aalling-Frederiksen, KMØ Jensen,; Design and Application of a Gas Diffusion Electrode (GDE) Cell for Operando and In Situ Studies ; Chimia 2024, 78, 344
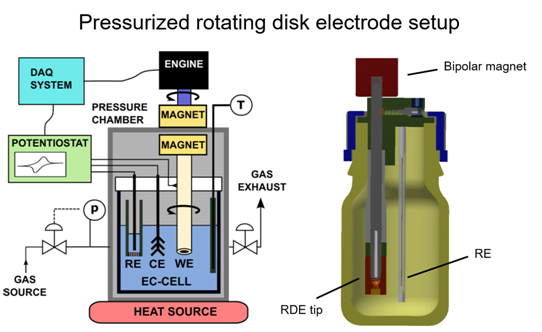
RDE setup for elevated temperature and pressure
In conventional rotating disk electrode (RDE) measurements, the accessible current and potential window for electrocatalytic investigations - such as oxygen reduction reaction (ORR) studies - is severely limited by the low solubility of gaseous reactants in the electrolyte. In addition, the applicable temperature range is restricted, further constraining kinetic analyses under realistic operating conditions.
To overcome these limitations, we developed a pressurized RDE system that significantly expands the achievable current density, potential window, and temperature range for kinetic studies. This setup enables investigations under elevated pressure and temperature, thereby providing improved control over mass transport and reaction conditions. For example, it has been successfully applied to studies in hot phosphoric acid environments relevant to high-temperature polymer electrolyte membrane fuel cells (HT-PEMFCs).
- ; Design and Application of External Reference Electrode for Kinetic Studies at Elevated Temperatures and Pressures;
- ; Rotating disk electrode system for elevated pressures and temperatures;
- ; Design and test of a flexible electrochemical setup for measurements in aqueous electrolyte solutions at elevated temperature and pressure;
